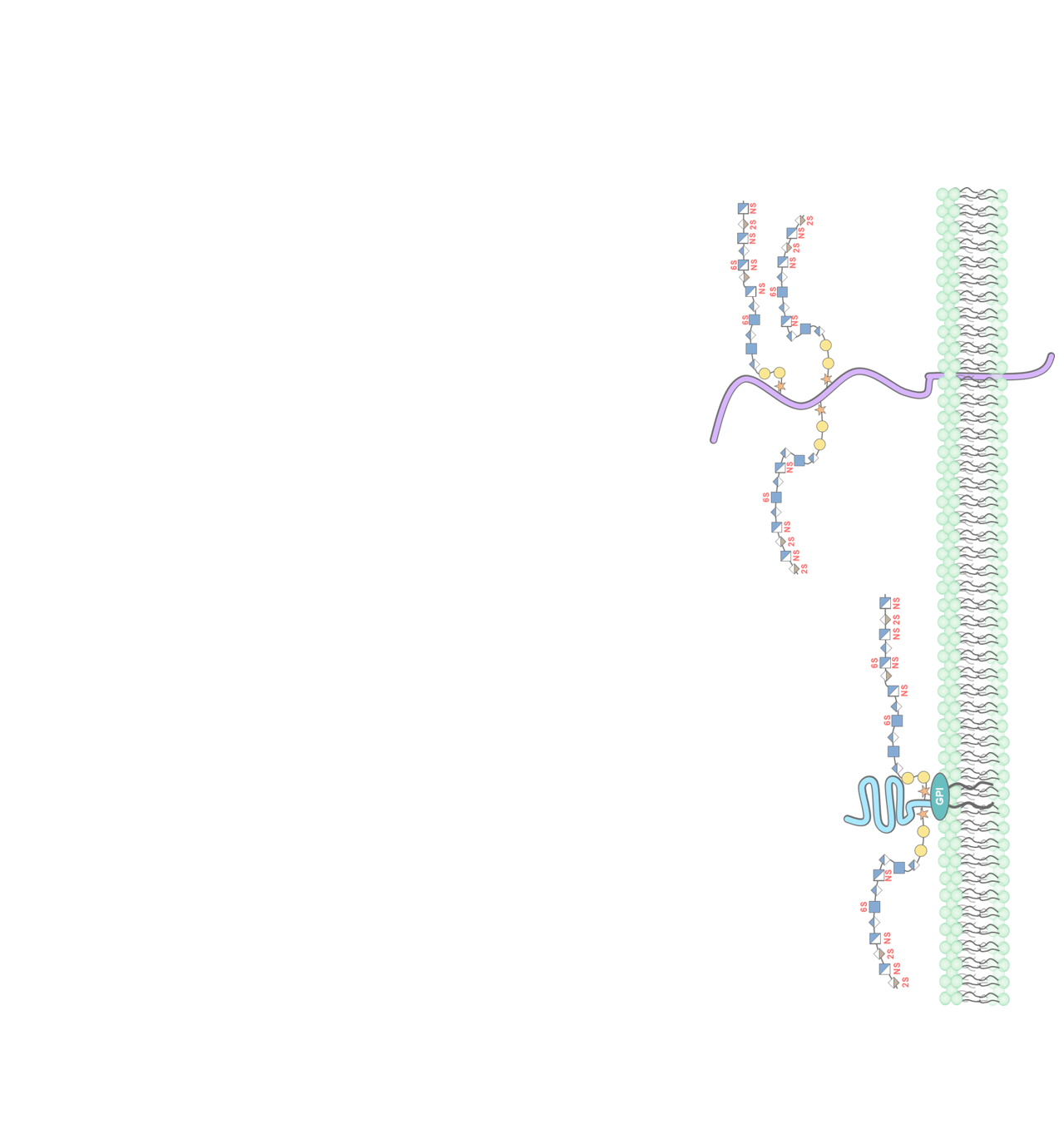
Glycosylation plays a variety of roles in basic biological processes, and alterations in these carbohydrate structures contribute to many human diseases. The mammalian glycome contains extensive structural and functional heterogeneity that can vary temporally and spatially during development and in different tissues. However, regulation of the assembly of these ubiquitous, non-template driven post-translational modifications is currently poorly understood. Research in the Weiss Laboratory aims to identify the molecular mechanisms responsible for genetic regulation of glycosylation in mammalian cells. In particular, we are focused on identifying the transcriptional and epigenetic programs responsible for temporal and spatial control of glycosylation using genome-wide, molecular, and genetic approaches. Our ultimate goal is to utilize these discoveries to identify novel drugs and drug targets for treatment of relevant human diseases.

Functional Genomics of Glycosylation
In the Weiss lab, we utilize pooled CRISPR screening, genetic engineering, and bioinformatic tools to identify the molecular mechanisms responsible for the genetic regulation of glycosylation.
Drug Discovery for Rare Genetic Disorders
Many human diseases are caused by rare mutations in genes involved in the biosynthesis and/or catabolism of glycans. In the Weiss lab, genetic engineering, high-throughput drug screening, and cell-based disease models are utilized to uncover novel drug targets and therapeutic agents to treat these rare genetic disorders.

Epigenetic and Transcriptional Regulation
Epigenetic changes alter the physical structure and organization of DNA, and transcription factors regulate the expression of their genomic targets. Currently, little is known about the genomic regulation of glycosylation. The Weiss lab aims to use state-of-the-art genomic tools and small molecule ligands to identify and modulate novel regulatory pathways responsible for controlling glycosylation in mammalian cells.